The Klotho gene has gained increasing attention for its anti-aging properties. In the most recent installment of this series, we explored the promising cognitive benefits of administering Klotho to both mice and monkeys, the results from which may be mirrored in humans. The benefits of this circulating hormone, however, extend beyond the brain.
Klotho was first discovered as the antiaging gene in 1997 when researchers found that enhancing its expression could increase the lifespan of mice by more than 30%. Although a variety of different genes and environmental factors can influence longevity, studies have shown that Klotho-deficient mice not only have shorter lifespans but also experience more age-related complications. Premature aging in these mice often was accompanied by loss of muscle and fat tissue, thinning skin, reduced fertility, cardiovascular complications, movement abnormalities, and bone disease. Since Klotho is primarily produced in the kidneys, it is not surprising that many of these age-related complications often result from kidney dysfunction.
The kidneys generate two types of Klotho: (1) a transmembrane protein that inserts itself into the cell membrane and mediates kidney function, and (2) a secreted hormone that is released into the bloodstream. Individuals with naturally high levels of the hormone in their blood seem to not only live longer and be more resistant to age-related complications but also perform better on learning and memory tasks. In fact, even when a relatively small dose of Klotho is administered, animal studies have shown that the brain undergoes significant changes that allow more connections to be made in the hippocampus, the brain’s learning and memory center.
A hormone as large as Klotho, however, cannot cross the blood-brain barrier. Instead, it relies on intercellular signaling. A team from the University of California San Francisco found that Klotho encourages glutamate signaling in the NMDA learning pathway. In the presence of glutamate, NMDA proteins activate to promote synaptic plasticity, a process that is integral for learning and memory. Klotho circulating in the blood seems to trigger the release of glutamate across the blood-brain barrier, to such an effect that it continues to provide long-lasting benefits even once the hormone is cleared. This not only protects against cognitive decline but may also reverse it.
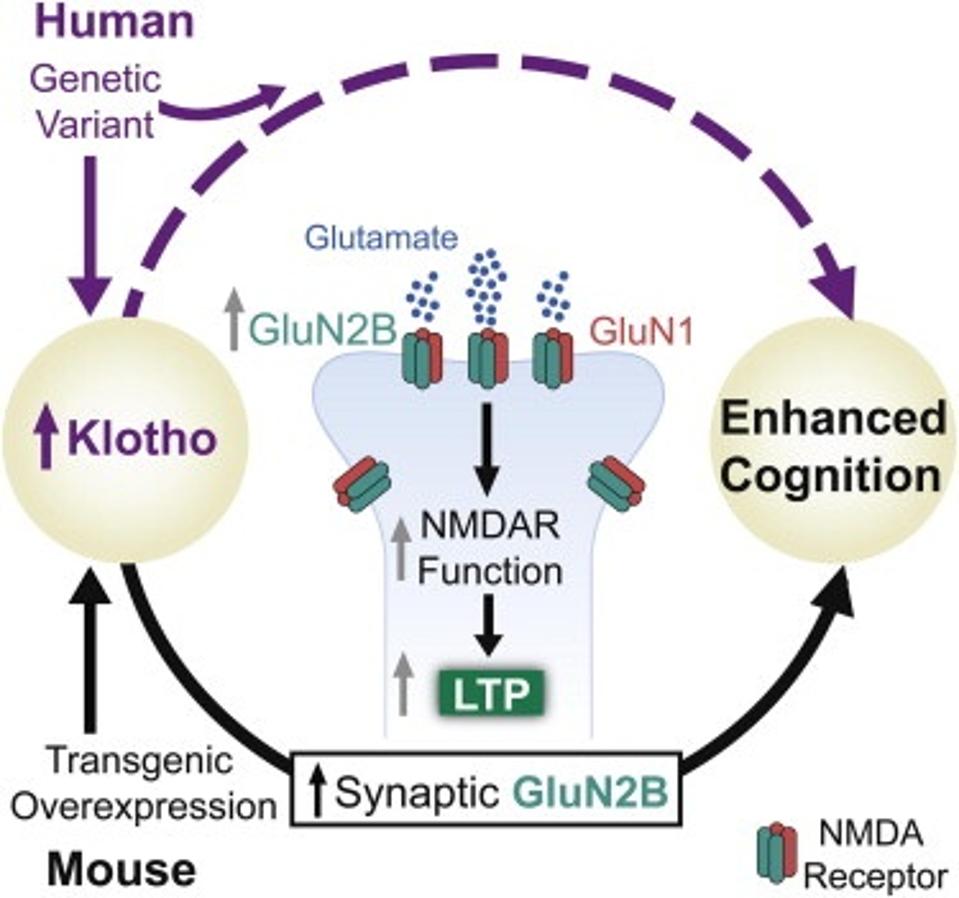
In the presence of glutamate, NMDA proteins activate to promote synaptic plasticity, a process that is integral for learning and memory. Klotho circulating in the blood seems to trigger the release of glutamate across the blood-brain barrier, to such an effect that it continues to provide long-lasting benefits even once the hormone is cleared.
FROM: “LIFE EXTENSION FACTOR KLOTHO ENHANCES COGNITION” DUBAL ET. AL 2014
Beyond learning and memory, the mechanism through which Klotho is able to resist other age-related changes has been largely unknown until now. In their recently published study, researchers in China found that introducing Klotho into the bloodstream reduces inflammation and oxidative stress in both the kidney and the brain. Given that inflammation is one of the primary drivers of age-related complications, their findings suggest that the Klotho gene may be a promising therapeutic target for treating chronic inflammation, not to mention helping individuals live longer.
This study recruited a cohort of eight-month-old mice, equivalent to approximately forty-year-old humans. Compared to the control mice that were given saline injections, the Klotho-enhanced group had considerably lower levels of inflammation and oxidative damage in their kidneys. Although the hormone injections did not directly increase the amount of Klotho found in the brain, these mice were also more resilient to age-induced neuroinflammation and performed better on a series of water maze tasks that measured learning and memory.
To understand the impact of Klotho at the cellular level, investigators also obtained a RAW264.7 tissue sample, known to express a particular type of inflammatory cell called macrophages. Macrophages are a type of white blood cell that are recruited to fight infections. These cells can take on two forms. M1 macrophages release proinflammatory factors to kill foreign pathogens, but these toxins can also damage otherwise healthy cells. Once the pathogen has been eliminated, signaling changes trigger the conversion of M1 macrophages into their M2 subtype. M2 macrophages are then responsible for repairing and regenerating injured tissue. In chronic inflammatory conditions, investigators speculate that the effect of M1 macrophages is prolonged, preventing their conversion into their M2 subtype.
To their surprise, Wang et. al found that exposure to Klotho significantly reduced the number of pro-inflammatory M1 macrophages and enhanced the activity of anti-inflammatory M2 macrophages. These findings were mirrored in both the brains and kidneys of the Klotho-enhanced mice. Investigators concluded that the Klotho hormone stimulates a signaling pathway that triggers this conversion, though the exact mechanism is not yet clear.
Although the role of Klotho in mediating inflammation remains an active field of study, the possible therapeutic effect is promising. Understanding the role of this protein may hold the secret to preventing aging. A future in which Klotho hormones can be packaged and sold supplements may be closer than we think.


