The current generation of monoclonal antibodies are ineffective against the predominant variants of SARS-CoV-2 in circulation. Recent studies by Callaway et al. open the possibility that a new type of monoclonal antibody may regain some of the lost activity. These are bivalent antibodies that recognize two sites on the virus simultaneously.
From the early days of the pandemic, the SARS-CoV-2 virus began to mutate. Over the following years, entire new lineages of variants emerged, such as Alpha, Beta, Gamma, Delta, and now Omicron. Each lineage introduced new mutations that altered the virus’s structure and enhanced its ability to infect host cells and evade neutralizing antibodies.
Omicron and its sublineages are the current exemplars of antibody evasion. Vaccines and antibody treatments used against earlier variants had greatly reduced effect on the Omicron family. There are some antibodies, however, that maintain some, if not all neutralizing capacity against the new viruses, namely bivalent antibodies.
Antibodies that recognize the Omicron spike bivalently attach to two receptor binding domains on the spike trimer, as opposed to one. Most monovalent antibodies fall short against Omicron, but bivalent antibodies show promise. Researchers Callaway et al. examined the neutralizing capacities of nearly 400 antibody therapeutics to further understand the supremacy of bivalent antibodies. Here we discuss their results and the further implications on vaccine and treatment design.
The 397-Antibody Panel
The researchers introduced a massive panel of nearly 400 antibodies to a wide variety of viruses, including D614G, Beta, Delta, Mu, and three sublineages of Omicron. At a low concentration of 250 ng/mL, only 66 potently neutralized BA.1, the base of the Omicron tree. At the higher 25 μg/mL, an additional 67 antibodies neutralized BA.1.
The researchers further tested 20 antibodies that potently neutralized the full panel against BA.4/5 and BA.2.12.1, later strains of Omicron known for their additional mutations and antibody resistance. The 20 maintained potent neutralization of Ba.2.12.1, but 14 had reduced or complete loss of neutralization against BA.4/5. The authors attributed this to the F486V receptor-binding domain mutation unique to BA.4/5. Most notably, the remaining antibodies neutralized the Omicron sublineages as well or better than the D614G parent lineage.
Bivalent Binding
Upon further examination using electron microscopy, Callaway et al. found that of the Omicron-neutralizing antibodies, 13 bound bivalently, with each arm of the antibody engaging a neighboring receptor-binding domain in the spike. An IgG antibody is ‘Y’ shaped, with two arms and a base. The arms are those that bind the spike protein.
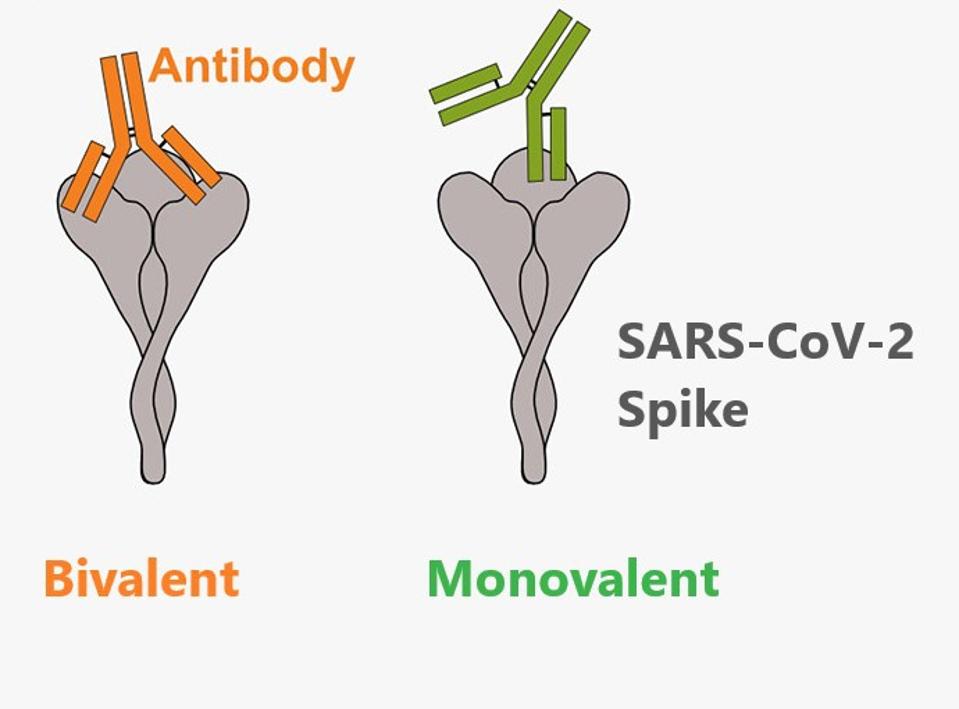
FIGURE 1: Bivalent vs monovalent binding of an antibody to the SARS-CoV-2 Spike protein.
CALLOWAY ET AL.
The researchers confirmed the connection between bivalency and antibody-spike affinity by conducting the same electron microscopy with nine antibodies that failed to neutralize Omicron. Eight of the nine bound partially or entirely monovalent, suggesting a link between bivalency and Omicron neutralization.
Impact of Bivalency on Neutralization
Callaway et al. further investigated the supremacy of bivalency by isolating the Fab fragments of the bivalent Omicron-neutralizing antibodies. Essentially, they tested one arm of the ‘Y’ as opposed to the entirety of the ‘Y’ to deduce the importance of the arms working in tandem.
Using eight bivalent potent antibody fragments, they found that all had lower neutralization than intact IgG antibodies against D614G. All eight had an even more precipitous drop against Omicron BA.1 pseudovirus.
Discussion
The disproportionate effectiveness of bivalency in Omicron neutralization may be a marker for antibody and vaccine design in the near future. For over a year, Omicron sublineages have dominated SARS-CoV-2 virus frequencies. Given the widespread nature of the Omicron family, this may be unlikely to change anytime soon. If bivalent antibodies are those prone to bind and neutralize Omicron viruses, perhaps we should focus efforts on the discovery and isolation of bivalent antibodies for treatment and vaccines.
Bivalent antibodies are far from a new concept. For instance, the discovery of bivalent virus-like particles geared towards Ebola has led to recent advancements in the search for additional effective Ebola vaccine candidates. A bivalent vaccine for Respiratory Syncytial Virus also takes a similar approach.
With bivalency as a prerequisite for Covid-19 antibody treatments, we may conduct a more thorough search of broadly neutralizing antibodies in development, bringing forward those with bivalent tendencies. Omicron and its many descendants will likely remain in the public sphere for some time. Bivalent antibody treatments would be another arrow in the quiver to keep the Omicron family at bay.