Recent studies indicate the previously unknown protective value of non-neutralizing monoclonal antibodies against SARS-CoV-2. Throughout the pandemic, the search for monoclonal antibody treatments centered around neutralizing capacity. How well can an antibody neutralize the latest variants? As new variants arose, previously neutralizing antibodies lost their potency.
In a previous description, we analyzed the discovery of non-neutralizing antibodies protecting humanized mice models from Covid-19. In fact at proper dosages, some non-neutralizing antibodies protected better than their neutralizing counterparts. The key was opsonization: the tagging of pathogens for elimination by phagocytes. Non-neutralizing antibodies efficiently opsonized, leading to rapid clearance of the virus from infected subjects.
Here we discuss a study by Izadi et al., which expands on the promise of non-neutralizing antibodies, as well as the potential of antibody subclass switches to enhance immune functions.
The two main components of an antibody are the Fab fragment and the Fc fragment. The Fab is the section that binds the pathogen and modulates neutralization. The Fc fragment binds host cell Fc receptors and modulates immune activities. Most human monoclonal antibodies isolated from sera are IgG1 antibodies. Neutralizing capacity aside, IgG1 is the joint-most potent activator of Fc gamma receptors on immune cells, alongside IgG3. However, IgG3 is the single most potent activator of the classical complement pathway, which mediates immune functions such as inflammation, opsonization, and pathogen clearance.
Izadi et al. reengineered eight high affinity and non-neutralizing SARS-CoV-2 antibodies to use an IgG3 structure as opposed to an IgG1.
FIGURE 1: Schematic of the heavy and light chain plasmids containing the variable and constant domains.
IZADI ET AL.
For six of the eight, the altered Fc fragment did not impact the binding affinity of the antibodies to the SARS-CoV-2 virus, meaning the enhanced immune functions of IgG3 did not come at the cost of binding capacity. For the remaining two antibodies, altering the Fc fragment substantially altered avidity. In one case, Ab11, the binding affinity for IgG3 was far worse than IgG1, yet for Ab57, IgG3 far outperformed IgG1, suggesting that Fc class switching can have an impact on avidity, but to exactly what degree remains unknown.
IgG3-switched antibodies also induced a stronger phagocytosis response than their IgG1 counterparts. Phagocytosis is the ingestion and elimination of pathogens by immune system phagocytes. Opsonization is the flagging of these cells to destroy an invader. All antibodies, aside from Ab11, saw phagocytosis efficiency increase up to threefold.
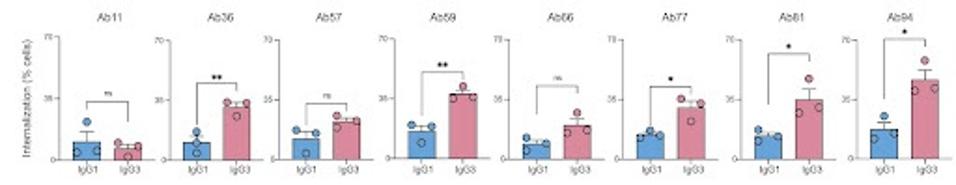
FIGURE 2: IgG3 antibodies are superior to IgG1 antibodies at inducing Fc-mediated phagocytosis further potentiated when used as oligoclonal cocktails.
IZADI ET AL.
Furthermore, cocktails of the antibodies improved phagocytosis even further. A cocktail of all eight IgG3 antibodies was 12-fold more potent at phagocytosis induction than a cocktail of their IgG1 counterparts. A dual cocktail of the top two IgG3 antibodies was 8-fold more potent than its IgG1 counterpart.
As for complement activation, IgG3 completely outperforms IgG1, which failed to even activate the system. Ab94, the best performing singular IgG3, was the only single antibody that could induce complement deposition. The dual cocktail and octo-cocktail each deposited roughly 2.5-fold more than Ab94. Taken together, IgG3, and more specifically IgG3 antibody cocktails more effectively trigger the complement system, which would lead to more rapid clearance of the virus in a patient.
Finally, Izadi et al. aimed to validate their findings in live models to determine the relevancy of Fc mediated functions against true SARS-CoV-2 infection. Recall from our previous description that these antibodies do not have neutralizing capacity, so any and all protective capability is dependent upon Fc mediation. They prophylactically injected mice with Ab94 IgG1, Ab94 IgG3, Ab81 IgG3, DuomAb IgG3 or a vehicle control, followed by infection with the Wuhan strain of SARS-CoV-2.
All mice inoculated with some form of antibody outperformed those in the control group, in terms of body weight, mortality, and lung viral load. Notably, the Ab94 IgG1 performed the best in all areas, but the IgG3 antibodies and IgG3 cocktail were not far behind. This data adds to the growing evidence that both Fc-mediated immune functions are crucial to antibody development, as well as the value of non-neutralizing antibodies as treatment avenues.
Discussion
The use of these highly engineered antibodies for increased opsonization may be the recipe that’s needed to revive antibody therapy to revive antibodies as effective treatments for a broad range.
One caveat of note is that of resistance. These antibodies mostly bind the receptor-binding domain, aside from Ab94, which binds both receptor-binding and N-terminal domains. This opens up the possibility of virus mutations impacting the binding affinity of these non-neutralizing antibodies, which could make them less effective.
The search for more such antibodies not only that S1 subdomains, but also antibodies such as the camelid antibodies and others bind mind multiple faces of the trimeric structure to lock the spike from fusion. This style of antibody worked to improve the neutralizing activity for other viruses and we suggest coupling that with the major advance here of increasing opsonization by IgG3 FC fusion. A combination in that model offers a way forward for resurrecting monoclonal antibodies as effective treatments for a wide range of variants both past and future.