|
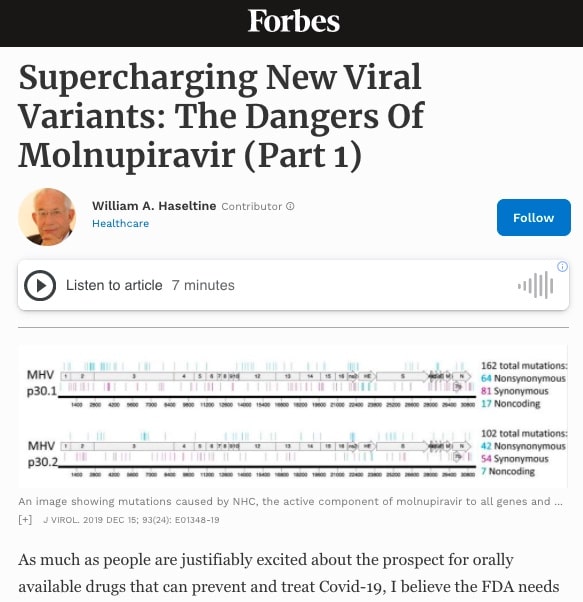
| As much as people are justifiably excited about the prospect for orally available drugs that can prevent and treat Covid-19, I believe the FDA needs to tread very carefully with molnupiravir, the antiviral currently before them for approval. My misgivings are founded on two key concerns. The first is the drug’s potential mutagenicity, and the possibility that its use could lead to birth defects or cancerous tumors. The second is a danger that is far greater and potentially far deadlier: the drug’s potential to supercharge SARS-CoV-2 mutations and unleash a more virulent variant upon the world. My next two articles will explore both of these concerns in much greater detail. But let me be very clear right from the start: I am a strong believer in antiviral drugs in general as a means to control the pandemic, having spent much of my early career focused on developing antivirals for the world’s last major pandemic, HIV/AIDS. But I have also spent many years — at Harvard especially where I founded and chaired the Division of Biochemical Pharmacology — studying mutagenesis and the long term effects of damaged DNA. My concern with molnupiravir is because of the mechanism by which this particular drug works. Molnupiravir works as an antiviral by tricking the virus into using the drug for replication, then inserting errors into the virus’ genetic code once replication is underway. When enough copying errors occur, the virus is essentially killed off, unable to replicate any further. The FDA will soon be debating the safety of molnupiravir for high-risk individuals with Covid-19, something which I will explore in greater detail in my next piece. But my biggest concern with this drug is much larger than the health of any one person, it is molnupiravir’s ability to introduce mutations to the virus itself that are significant enough to change how the virus functions, but not so powerful as to stop it from replicating and becoming the next dominant variant. In a series of pre-pandemic experiments to determine whether coronaviruses could become resistant to molnupiravir (the answer: yes, they can), researchers tested the active form of molnupiravir against two other highly pathogenic coronaviruses: MERS-CoV and the mouse hepatitis virus (MHV). To identify mutations associated with these phenotypes after passage, the authors sequenced complete genomes of two MHV lineages and two MERS lineages. With MERS, there were up to 41 mutations scattered across the genome (see Figure 1). With MHV, there were more than 100 mutations which occurred at every part of the genome (see Figure 2). |
 |
 |
| Overall, the study showed a dose-dependent increase in mutations for both coronaviruses, including in the all important spike protein which is the focus of so much attention today in SARS-CoV-2 variants of concern, like Delta. Critically, the researchers found that the viruses could survive and replicate to high titers despite such large numbers of mutations in every gene and protein. The viruses tested did show a slight replication disadvantage — though they still replicated to the same high titers, they did so slightly less rapidly compared to the original non-mutated viruses. However, outside of the lab, as the drug is given to millions of people with active infections, this disadvantage may quickly disappear as we would likely provide a prime selection environment to improve the fitness of the virus. While it’s possible that at the optimal concentration, the drug may very well cause enough mutations to prevent replication and onward transmission of the virus, the impact of suboptimal doses is still very much unknown. The current protocol for the use of molnupiravir is an 800mg dose, given as pills, twice a day for five days. At that concentration, molnupiravir would theoretically take no prisoners, leaving not a single viral genome to escape unscathed. But there is a strong likelihood that in the real world, people will not take the full course of pills. A slew of studies on adherence to daily oral antibiotics suggest that many patients — as many as 40% — fail to complete the full course of treatment. At these suboptimal concentrations, molnupiravir could have the unfortunate effect of introducing mutations across every gene and protein of the virus, including the spike, but not necessarily killing it off. The drugmakers, Merck and Ridgeback, as well as the FDA are exploring whether molnupiravir is safe for personal use in high-risk individuals with mild to moderate disease and whether its benefits outweigh any potential risks. But they should also be determining the broader danger, and how to prevent the drug from unleashing new and deadlier variants across the globe. Already SARS-CoV-2 has shown a remarkable ability to mutate and survive under pressure. The drug’s manufacturers, Merck and Ridgeback, are entering into licensing deals that would allow the drug to be made and sold widely in more than 105 countries, which means that, if approved by regulators, we will soon have very little control over the drug’s administration and dosages delivered. We are potentially headed towards a world class disaster. If the FDA does grant approval for the drug — and there is an argument to be made that better and safer antivirals are already on the way — it should be on a very narrow basis and include a black box warning to emphasize the potential danger of using the drug at suboptimal doses or for large numbers of people for preventive purposes. What we know with certainty is that his drug is far from the magic bullet we might hope for with an antiviral for Covid-19. The next article in this two part series will explore the potential risks and dangers of the drug for patients themselves. |