
This story is part of a series on the current progression in Regenerative Medicine. This piece discusses advances in regenerative devices and technologies.
In 1999, I defined regenerative medicine as the collection of interventions that restore to normal function tissues and organs that have been damaged by disease, injured by trauma, or worn by time. I include a full spectrum of chemical, gene, and protein-based medicines, cell-based therapies, and biomechanical interventions that achieve that goal.
A future where microscopic machines traverse the human body monitoring for vital signs and disease may be much closer than you think. What was once a facet of science fiction is now a reality as new ingestible vitals-monitoring pills complete their first in-human trials.
In a study for Cell Device, Dr. Giovanni Traverso and colleagues from the Massachusetts Institute of Technology describe a new biosensor they call the VM pill. The ingestible capsule contains sensors, a microprocessor to gather information, and a radio to transmit information back to a physician. But how could such a pill be used to help people? Here, I will analyze their invention and speak to its use in a regenerative medicine context.
As Traverso and colleagues note, ingestible electronics are far from new technology. The first trials of such devices date back to the 1950s, tracking simpler measurements such as temperature and pH balance. The VM pill, however, is far more advanced.
The device is 25 mm long, or about the size of an adult’s multivitamin. Within a sealed plastic shell lie two batteries, a processing unit, an accelerometer, and an antenna-equipped radio. On the receiving end of the signal is an antenna that feeds into data-monitoring software.

FIGURE 1: (B) VM pill, a vitamin-sized (9 × 25 mm) ingestible physiological monitoring device with a sealed biocompatible plastic shell. (C) VM pill components: 2 silver oxide batteries, an antenna, and 2 printed circuit boards (PCBs) shown in a 000 gel capsule for ease of visualization of internal components.
The researchers discuss two possible use cases for the device. The more speculative of the two is opioid overdose detection. For those struggling with opioid addiction or those being prescribed heavy sedatives, the VM pill could be used in tandem on a consistent basis to monitor for respiratory depression as a result of opioid overdose. Upon detection, the VM pill could notify first responders, akin to a life-alert-style button device designed for the elderly.
They confirmed the VM pill’s detection capacity by a controlled swine test in which a subject was given 250 micrograms of fentanyl, at which point the VM pill immediately detected the overdose due to decreased breath. The subject was then given a 1 mg dose of Naloxone and rebounded, at which point the VM pill also detected recovery of breath.
The second use is one they explore more significantly with their own trial: sleep study. Traditional standard-of-care sleep observation requires sleeping in a medical lab, attached to a series of wires, resulting in uncomfortability and inconsistent results.
Sleep study with the VM pill could allow for at-home observation while maintaining a more consistent sleep setting during data collection.
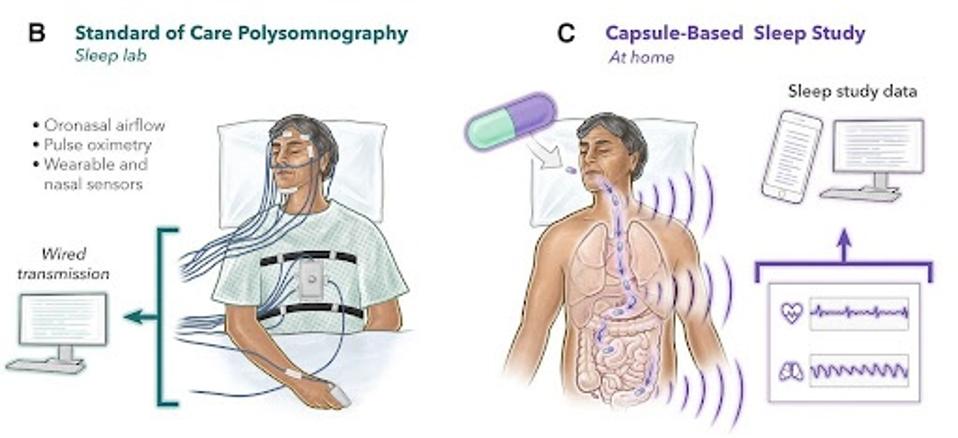
FIGURE 2: (B) Standard of care for polysomnography. (C) Capsule-based sleep study evaluation.
Comparing the accuracy of the VM pill to that of the wired polysomnograph, Traverso and colleagues found that the pill was nearly as accurate as the standard of care monitoring system. Respiratory rate accuracy was within one breath per minute of the in-lab system, and heart rate was within two to three beats per minute, both well within a reasonable margin of error. Further, the VM pill could also detect when patients were holding their breath or experiencing moments of sleep apnea. They also found that the VM pill maintained accuracy even when patients with sleep apnea used their ventilators.
What, then, are the limitations of the VM pill? Two that immediately come to mind are cost and length of use. To address the latter, the VM pill can only be used while in transit in the body, or roughly 36 hours on average. This limits long-term monitoring use cases.
Furthermore, depending on the price per pill of the device, patients will have to retrieve it after it makes its way through the digestive tract, which is likely to limit enthusiasm for the device. If the pill is relatively cheap, given its size and simple design, it could be prescribed to the patient in bulk, akin to normal pills, but no cost estimates were provided in the study.
Aside from respiratory and heart rate monitoring, future applications for the pill could come from alterations in its design, leading to other avenues of health monitoring. These may include digestive health, blood sugar monitoring, cancer cell detection, etc. I look forward to seeing how the system is adapted and implemented in the coming years.
To read more of this series, please visit www.williamhaseltine.com


