This story is part of a series on the current progression in Regenerative Medicine. This piece discusses advances in brain-computer interfaces.
In 1999, I defined regenerative medicine as the collection of interventions that restore to normal function tissues and organs that have been damaged by disease, injured by trauma, or worn by time. I include a full spectrum of chemical, gene, and protein-based medicines, cell-based therapies, and biomechanical interventions that achieve that goal.
New cortical brain-machine interface technologies will soon aid those with brain injury or neurodegenerative diseases to improve basic motor function. The motor cortex is the region of the cerebral cortex that controls all voluntary movement. In patients with brain injury or neurodegenerative diseases, of which there are tens of millions in the United States alone, sometimes the motor cortex is damaged, leaving the patient with reduced or a complete lack of motor function.
In an article for Current Biology, Dr. Richard Andersen and colleagues from the California Insitute of Technology describe using brain-machine interfaces to help this population. As I have discussed in previous articles, brain-machine interfaces are the communication between the brain and electronics via the brain’s electrical signals. Andersen describes recent advances in cortical prosthetics using brain-machine interfaces. Here, I will comment on Andersen’s findings and speak to their potential applications in the near future.
In healthy adults, the motor cortex will send electrical signals through the nervous system’s neural pathways to the intended muscle groups, which contract or relax when they receive the brain’s message.
For patients with damaged nervous systems, the message may still be created by a healthy motor cortex, but will not reach its intended target due to some disruption along the way. Most cortical brain-machine interfaces will feature an implanted chip in the patient’s motor cortex, which can record and translate neural activity to external devices, typically a prosthetic or accessory of some sort, which take the place of the contracting or relaxing muscle group and move the intended body part.
Andersen and colleagues focus on four topics regarding the above process: (1) expanding neural source controls, (2) brain signaling, (3) somatosensory feedback, and (4) decoding processes. We will examine each of these and discuss them.
Expanding Neural Source Controls
While the motor cortex is the primary target for sourcing neural signals for brain-machine interfaces, Andersen and colleagues note that other areas of the brain can also be used for neural connections.
For instance, the posterior parietal cortex is an area of the brain that connects the motor and sensory regions of the brain. Studies regarding the posterior parietal cortex have found that this region of the brain plays a role not in the movement itself but in the intentionality behind the movement. When we move our arm, there is often a motive, whether reaching, grasping, pointing, etc.
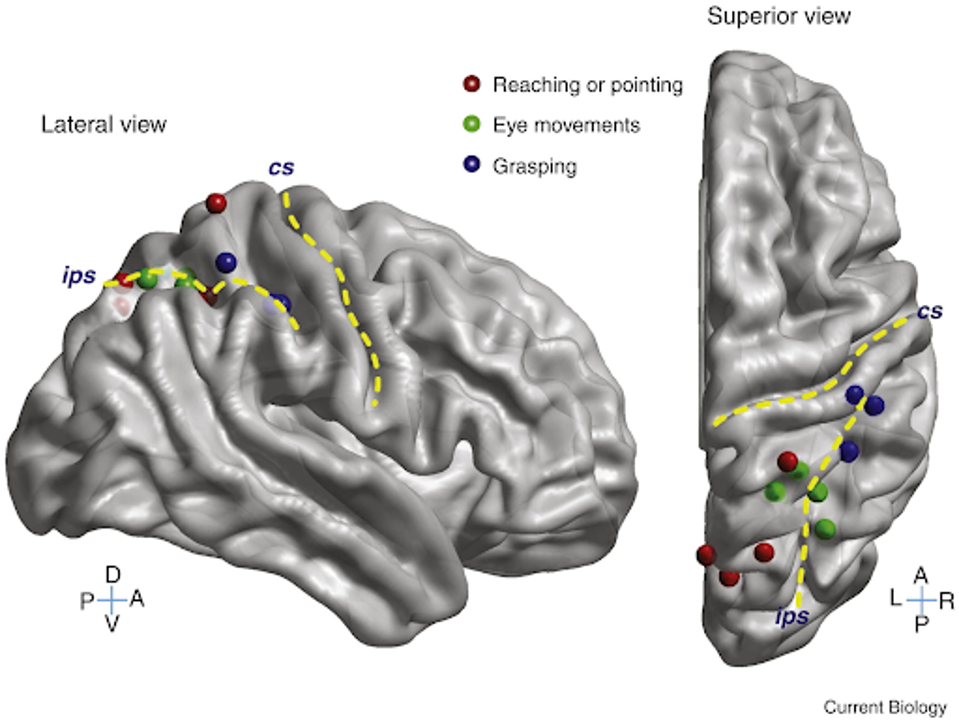
FIGURE 1: Organization of intent in the human posterior parietal cortex.
For prosthetic applications, the intention behind movement could be just as important when designing a brain-machine interface. Patients who use such a prosthetic would likely much rather be able to think, “I’m grabbing this cup,” than “squeeze my fingers around the cup.” Were the posterior parietal cortex to be implemented into the brain-machine implant, the entire system could be much more functional and even elegant.
Brain Signaling
When I say that brain-machine interfaces use neural signals to transmit information, there are actually many different options of neural signals to choose from. However, different neural signals require different levels of invasive electrodes in the brain implant, prompting questions of safety concerns. Single neuron firings may be more accurate for the prosthetic but would require a more significant implant, and the opposite is the case for larger-scale brain signals.
Andersen and colleagues note one type of signal that may represent the Goldilocks choice. Local field potential signals represent the collective activity of a group of neurons. They capture the brain activity of a larger area than it actually occupies, as though they take a picture of a larger specific brain area.
Local field potential signals contain information on motor tasks, such as arm movements and speech activities, but also more abstract information on intention, planning, and goals behind movements. If harnessed, local field potential could be a remarkably efficient signal for brain-machine use.
Somatosensory Feedback
One of the greater challenges with brain-machine prosthetics is the lack of sensory feedback during an action. A patient may see their prosthetic pick up an object, but the lack of feeling could be disappointing. I have discussed the advance of haptic feedback in recent articles, which may improve sensory feedback shortcomings in the near future, but cortical brain-machine interfaces could also take a different approach.
The solution would be a bidirectional brain-machine interface, which not only sends signals to the prosthetic from the brain but sends sensory information back to the brain from the prosthetic. Sensory information is collected by the nerves on our skin and sent to the somatosensory cortex of the brain. Sending specific artificial signals to the brain from the prosthetic could recreate the feeling of touch.
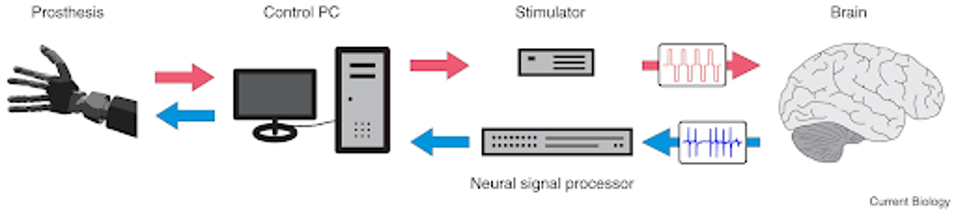
FIGURE 2: Schematic overview of a bidirectional brain–machine interface.
This would require including the somatosensory cortex in the brain implant, which is typically the limiting factor in these scenarios, as the larger the implant, the greater the risk of complication. Restoring the sense of touch would surely be monumentous to those who have lost touch sensation through disease or injury.
Decoding Processes
Perhaps the most difficult aspect of the brain-machine interface is the decoding of neural signals into actionable data. It is not as simple as converting ones and zeros from the brain to a computer program. Complex algorithms read the brain’s activity and translate the activity into data, but only after extensive training.
The decoder must be calibrated to the brain it is decoding. It is trained for dozens, if not hundreds, of hours to create reference points for the brain so that when it recognizes a certain signal, it can infer the action intended by the patient.
These signals are also not always the same. Our brain signals can change based on our mood, health, amount of sleep we’ve had, and a number of other factors. The decoder must take all this information into account when deciphering what the patient intends from their prosthetic.
Decoders are the limiting factor when discussing the upside of current brain-machine interfaces. As the decoder technology improves in terms of accuracy and efficiency, the viability of the interface increases drastically. Not to mention, as the technology progresses, the cost of such a system will decrease, making interfaces much more affordable for the millions of people suffering from brain injury or disease.
The rapid development of cortical brain-machine interfaces is one of the most exciting fields of study in modern regenerative medicine. The potential therapeutic, medicinal, and industrial applications of these devices is extraordinary. I highly anticipate the further development of such marvels as we venture into a new era of science and technology.
To read more of this series, please visit www.williamhaseltine.com