The Burden of Respiratory Syncytial Virus and the Need for a Vaccine
This story is Part I of a series on respiratory syncytial virus vaccines. Here we describe the disease itself, and in the next section, we will describe the steps forward towards producing the first safe and effective vaccines.
The success of the COVID-19 vaccines has reignited enthusiasm for the discovery of vaccines targeting diseases that have plagued us for many decades. In this series, we are going to discuss respiratory syncytial virus as a disease and why a vaccine is so badly needed. In the next section, we will describe the new research that is bringing us much closer to successfully producing the first safe and effective respiratory syncytial virus vaccines.
Respiratory syncytial virus is a highly contagious disease that causes severe respiratory illness in young children, the elderly, and the immunocompromised. It is one of the leading causes of death for infants globally, and a safe vaccine has yet to be approved for use. Furthermore, the one drug approved to prevent infection is effective only for a short period of time. That may change soon thanks to new discoveries about what went wrong with the very first vaccines and new insights into the structure of the virus that show a path forward.
Killing on average over 100,000 children across the world per year, respiratory syncytial virus (RSV) is the second greatest cause of death by a single pathogen for children under one year of age, falling behind malaria alone. Developing countries see an incidence of severe disease at double the rate of developed countries and account for an astounding 99% of the total deaths from RSV globally. This appalling disparity is in part due to the lack of resources to provide basic supportive care, which is why a reliable vaccine that provides lasting protection against this dangerous respiratory pathogen would make a significant difference in relieving global mortality.
Even in high income countries such as the United States, respiratory syncytial virus persists as a serious health burden, as it is the leading cause of hospitalization of children younger than five. As Jha et al. from Imperial College’s Respiratory Sciences, National Heart and Lung Institute reports, 50% of these hospitalizations occur in children under 6 months of age, demonstrating how infants in their first few months of life are particularly unequipped to fight off the virus. 10% of cases in children under five require hospitalization, and risk factors such as low birth weight, prematurity, and congenital lung or heart disease result in much higher rates of hospitalization. For older, healthy individuals, RSV can present as mild upper respiratory tract infection symptoms, such as a runny nose, sneezing, and coughing. But in more serious cases, it presents as a severe lower respiratory tract infection requiring hospitalization and mechanical ventilation, often in the form of bronchiolitis or pneumonia.
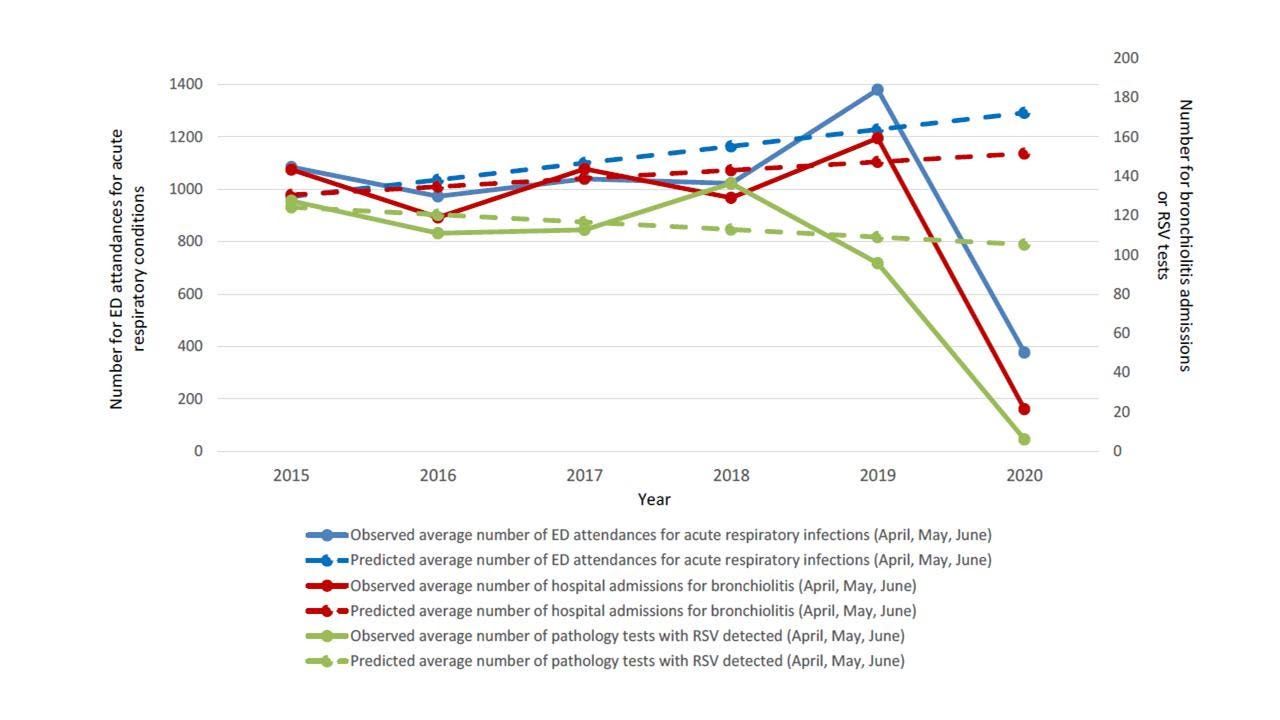
Figure 1. Observed and Predicted Trends of the Frequency of RSV PCR Detections, Bronchiolitis Admissions, and Emergency Department Acute Respiratory Visits. New South Wales (NSW), Australia, from 2015 to 2020 for children aged less than 16 years. (BRITTON ET AL.)
Also at additional risk for severe disease from RSV are the elderly and immunocompromised. Published in the Journal for Infectious Diseases, Moyes et al. found that HIV-infected children were found to be 3.5 times more likely to be hospitalized with RSV acute respiratory infection. Because respiratory syncytial virus infects approximately all children by the age of three and 60-70% by the first year of life, virtually all immunocompromised children are at risk. However, it doesn’t stop there. Because natural immunity from RSV infection doesn’t last beyond a few months, older children and adults continue to be reinfected throughout their lives. Although this doesn’t pose a threat for most healthy adults, those undergoing chemotherapy, organ transplant, or with underlying conditions such as chronic heart and lung disease are at a greater risk for serious health effects. Because the virus is so prevalent, it is hypothesized to have an adverse impact on the health of the elderly comparable to influenza.
The frequency of respiratory syncytial virus infections is seasonal, at least in temperate locations, and infections are much more common during the colder winter months. This adds to the higher burden hospitals face during the winter months, putting a strain on healthcare systems and increasing chances of co-infection with other respiratory viruses such as the common flu.
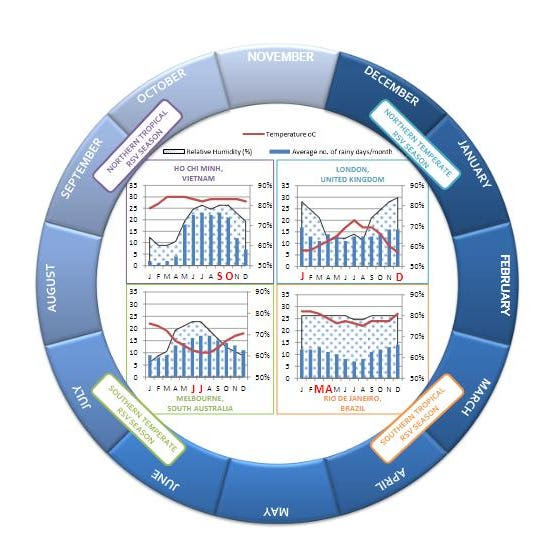
Figure 2. Annual Cycle of Global Peak RSV Incidence and Associated Climatic Factors. Temperate countries typically have peak incidence of RSV during winter months, which are associated with lower temperatures, higher rainfall, and higher humidity. There is greater variability in tropical regions, which are prone to biennial and perennial RSV. Limited information is available for some regions.(JHA ET AL.)
Discovered and initially characterized from a colony of chimpanzees in 1956 and later isolated from several infants, respiratory syncytial virus was given its name because it causes cells to fuse together, creating a multinucleated syncytium, or a giant cell containing multiple nucleases. Similar to other respiratory viruses such as COVID-19, infection begins by contact with respiratory droplets to the eyes or nose. Its spread is encouraged by the extended survival rate of viral particles on skin, cloth, and other surfaces.
Currently, treatment for RSV consists of monoclonal antibodies. Created by cloning white blood cells, monoclonal antibodies work by binding to a specific part of the antigen targeted by the antibody. However, monoclonal antibody treatment is only available in limited circumstances and must be administered intravenously each month, making it an impractical and expensive universal treatment. Palivizumab, the only licensed monoclonal antibody drug for prevention of severe infection by respiratory syncytial virus, is only permitted for use in high-risk infants and often must be administered five separate times per high incidence winter season. An accessible, safe, and reliable preventative treatment, a vaccine, is direly needed. Respiratory syncytial virus has posed obstacles to the technology that has been successful in producing other functional vaccines, and a failed vaccine trial with a tragic outcome over 50 years ago cast a dark shadow over new research for many years. Until recently, that is.
Monoclonal Antibody Treatment(PHOTO BY CRAIG F. WALKER/THE BOSTON GLOBE VIA GETTY IMAGES)
An effective respiratory syncytial virus vaccine must provide greater immunity than that provided by natural infection without causing any of the harmful effects as seen in the first vaccine trial back in 1965. The answer lies in better understanding the structure of RSV, and recent research has provided serious reason for hope. In the next section we’ll tell you about the challenges with respiratory syncytial virus’s structural proteins that must be solved, the explanation behind the disastrous 1965 vaccine trial, and how there are now promising vaccine candidates on the horizon.