This story is part of a series on the current progression in Regenerative Medicine. This piece is part of a series dedicated to the eye and improvements in restoring vision.
In 1999, I defined regenerative medicine as the collection of interventions that restore tissues and organs damaged by disease, injured by trauma, or worn by time to normal function. I include a full spectrum of chemical, gene, and protein-based medicines, cell-based therapies, and biomechanical interventions that achieve that goal.
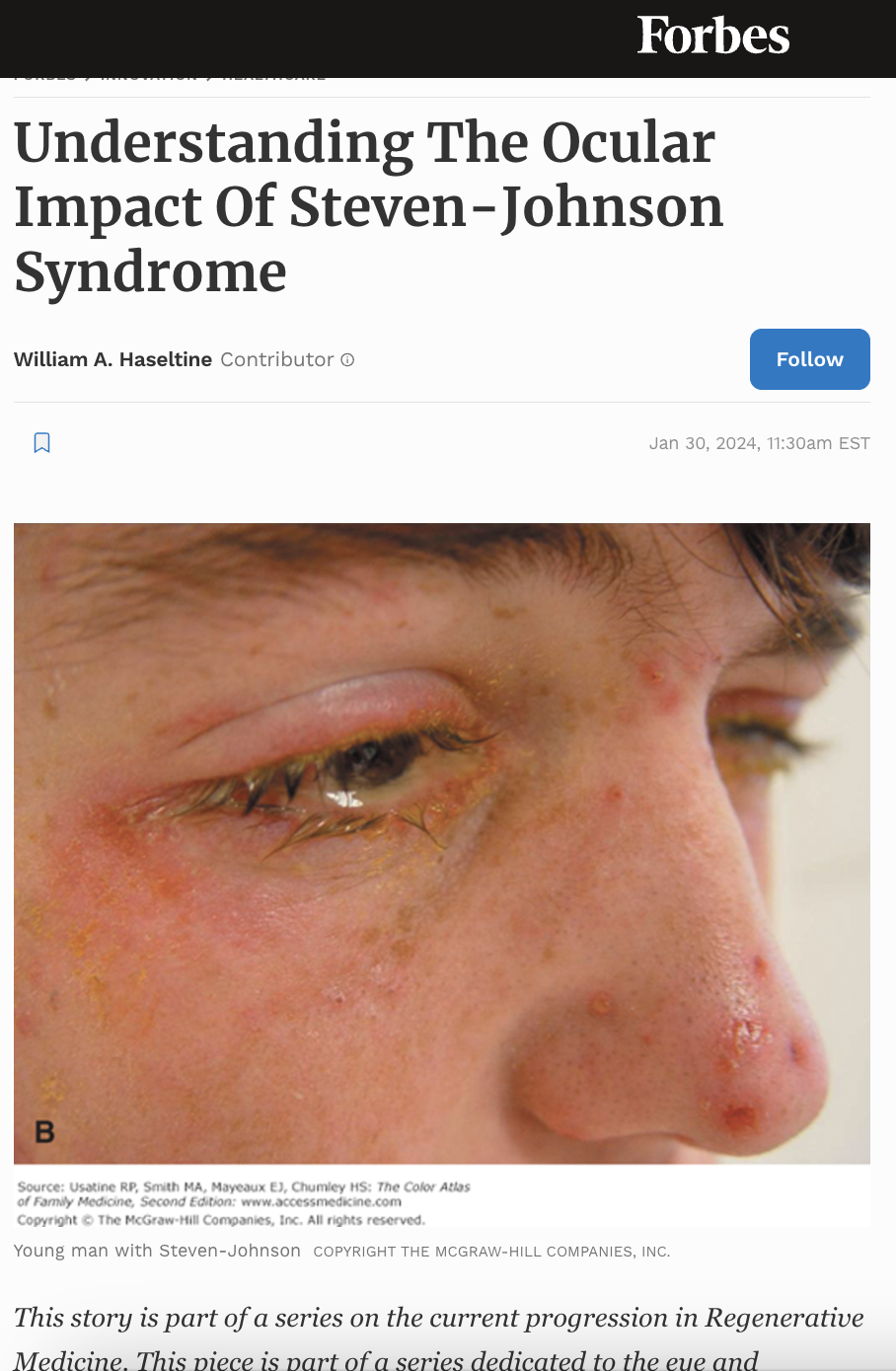
Steven-Johnson Syndrome (SJS) is a rare and life-threatening disorder that severely affects the skin and mucous membranes. SJS often results from an adverse reaction to medication or infection, causing detachment of the skin layers and the development of blisters and sores on the mucous membranes. The impact of SJS on the eyes is particularly prominent, resulting in severe eye pain, light sensitivity, vision loss, and, in some cases, blindness. However, advances in care and treatment options for SJS have vastly improved patient outcomes, including the ability to preserve eye health.
Novel treatments, such as intravenous immunoglobulin (IVIG) and systemic corticosteroids, have demonstrated promising results in treating SJS and reducing its associated ocular damage. The development of new treatments provides hope for SJS patients. Early intervention is critical to improving clinical outcomes and mitigating long-term effects.
Understanding Stevens-Johnson Syndrome and the Cornea
Stevens-Johnson syndrome (SJS) is a rare but life-threatening medical condition that can cause several ocular complications. The condition can affect people of any age, but it is most commonly found in children and young adults.
One of the most significant corneal complications associated with SJS is the development of fluid-filled blisters called bullae on the cornea’s surface. These bullae can cause considerable pain and discomfort, and their rupture can lead to corneal ulceration and perforation. Moreover, the rupture of these bullae can cause the cornea to become thin and fragile, increasing the risk of further complications.
SJS can also cause inflammation of the iris, the colored part of the eye. This inflammation can lead to iritis, which can cause eye pain, redness, and sensitivity to light. In some cases, SJS can also cause conjunctivitis, inflammation of the thin, clear tissue covering the white part of the eye.
This slit-lamp photograph showcases the enduring effects of SJS/TEN, encompassing limbal stem cell deficiency and symblepharon, among others.
In addition to bullae formation, SJS can cause significant damage to the corneal epithelium, the cornea’s outermost layer. This damage can lead to long-term changes in the corneal surface, including scarring and irregular astigmatism. Such changes can cause vision problems that persist even after the acute phase of the condition has resolved.
Treating of SJS and Corneal Complications
The primary treatment for SJS is to identify and eliminate the medication or other factors that caused it. Once the causative agent is removed, supportive care becomes essential to minimize the risk of complications and promote healing.
Supportive care may include pain management with nonsteroidal anti-inflammatory drugs (NSAIDs) or opioids, nutritional support, and fluids to prevent dehydration. Hospitalization is often required as SJS can rapidly progress and lead to life-threatening complications such as sepsis, organ failure, or pneumonia. In the hospital, patients are closely monitored and treated with intravenous fluids, antibiotics, and corticosteroids to reduce inflammation and promote healing.
SJS can cause severe damage to the cornea, which can lead to vision problems and even blindness if not treated promptly. Eye drops and ointments can manage pain and inflammation to protect the cornea from further damage and promote healing; bandage contact lenses can be worn. However, severe cases may require surgery to repair corneal perforation or remove damaged tissue.
Reviewing Clinical Trials and Outcomes
Researchers are conducting several clinical trials to explore new treatments for SJS and its associated ocular complications. These trials are focused on developing targeted therapies that can help reduce inflammation and promote healing.
There is a promising treatment for severe ocular complications caused by Stevens-Johnson syndrome (SJS). This treatment involves mesenchymal stem cells, multipotent stromal cells capable of differentiating into various cell types. Mesenchymal stem cells have anti-inflammatory and antioxidant properties that help corneal healing. Recent research has shown that using mesenchymal stem cells in a patient with SJS-induced corneal damage has significantly improved corneal clarity and reduced pain. This approach offers a potential solution for treating severe ocular complications associated with SJS.
Another promising treatment is a topical gel containing an anti-inflammatory agent and a neuroprotective peptide. In a small study, SJS patients treated with this gel significantly reduced ocular symptoms. The gel was safe and well-tolerated, with no adverse effects reported. This treatment may offer a viable alternative for SJS patients who are unable to tolerate traditional therapies.
Even though these trials are still in the initial stages, the outcomes have been promising. These studies bring hope to SJS patients who are experiencing severe eye-related complications and could create a path for more efficient treatments in the future. As the research progresses, the ultimate objective is to reduce or even avoid the harmful effects of SJS on the cornea.
To learn more about the eye, read more stories at www.williamhaseltine.com