Medical innovation Chimeric Antigen Receptor T cell (CAR T) therapy has attracted attention in recent years for its ability to send difficult-to-treat blood cancers into remission—even curing cancer for a minority. Now, research suggests that the therapy could potentially be adapted to treat a rare autoimmune disease called myasthenia gravis. The small clinical trial delivered a favorable safety profile and may even cause a long-lasting reduction in symptoms. A larger scale randomized control study is in progress and anticipated to finish around December of this year.
What is Myasthenia Gravis?
Myasthenia gravis is a rare, lifelong autoimmune disease estimated to impact 20 out of every 100,000 Americans. As with other autoimmune diseases, the immune system mistakenly attacks the body’s tissues. Myasthenia gravis is partly mediated by autoantibodies damaging communication between the neurons and the muscles, in particular. The disruption prevents the skeletal muscles which control the eyes, face, arms, legs and more from contracting as they should.
This mix of Latin and Greek words translates to “grave muscle weakness.” As the name states, people with myasthenia gravis lose control of certain muscles, manifesting tell-tale signs that include drooping eyes, unstable walking, as well as trouble talking and swallowing. Notably, physical activity worsens symptoms while rest usually improves them. Symptom management will usually entail medication to improve muscle weakness and suppress the immune symptom.
CAR T Therapy for Cancer vs Autoimmune Disease
CAR T therapy is a novel cell-based treatment which extracts and genetically modifies a patient’s immune cells to fight their cancer. Synthetic DNA is delivered to a set of immune cells called cytotoxic “killer” T cells. The DNA encodes a receptor which merges the T cell’s natural ability to destroy cancerous cells with honing abilities associated with antibodies. These permanently modified T cells become powerful, but hard to control, cancer killers upon infusion.
This model has yielded optimistic and long lasting results for many patients with certain lymphomas, leukemias, and multiple myeloma. Tantalized by this early success, researchers hope to tweak the platform to treat other illnesses, too. Examples of other efforts currently underway include CAR T therapy for lupus, cardiac fibrosis, and rheumatoid arthritis.
Autoimmune Context
Despite its promise, traditional CAR T therapy for cancer carries risks that are particularly unfavorable for people with myasthenia gravis.
Conventional CAR T cells cannot be controlled once infused. The engineered cells often overstimulate the immune system, causing two common yet potentially fatal adverse events: cytokine release syndrome (CRS) and neurotoxicity. This is disadvantageous to people with myasthenia gravis, who already have an overactive immune system and neurological symptoms; these effects can exacerbate already present complications.
Additionally, preparatory chemotherapy is usually required before an infusion of conventional CAR T cells. The short course of chemotherapy drugs destroys some immune cells to make room for the infused CAR T cells to proliferate later on. Further immunosuppression on top of already prescribed medication for myasthenia gravis could lead to an increased risk of infections and imbalance an already delicate immune system.
Descartes-08: CAR T Therapy for Myasthenia Gravis
In their study, Granit et al. turn to an alternative and temporary CAR T therapy called Descartes-08 to treat patients with myasthenia gravis. The therapy was originally designed for multiple myeloma patients but was later translated. This is possible because both diseases share a potential therapeutic target: B cell maturation antigen (BCMA) found on the surface of mature plasma cells. Cancerous plasma cells drive myeloma progression, and pathogenic plasma cells produce toxic antibodies that contribute to myasthenia gravis.
The most important feature, however, is the therapy’s half-life. It relies on synthetic mRNA, an inherently unstable molecule, to deliver the genetic information needed to encode the chimeric receptor. Unlike DNA, this information is not integrated into the cell genome. The synthetic mRNA naturally degrades in the T cell after a week, and receptor expression follows.
Theoretically, mRNA based CAR T cells could mitigate several risks associated with traditional CAR T therapy. Each infusion of T cells may elicit a weaker response, but they can be controlled through dose repetition; the slow and gradual introduction of T cells should prevent immune system overload. Likewise, lymphodepletion would not be needed to make space for these CAR T cells.
Study Details
The promising design needed to be tested. The researchers in this study sought to determine how safe the therapy was, and what dose would be ideal for treatment.
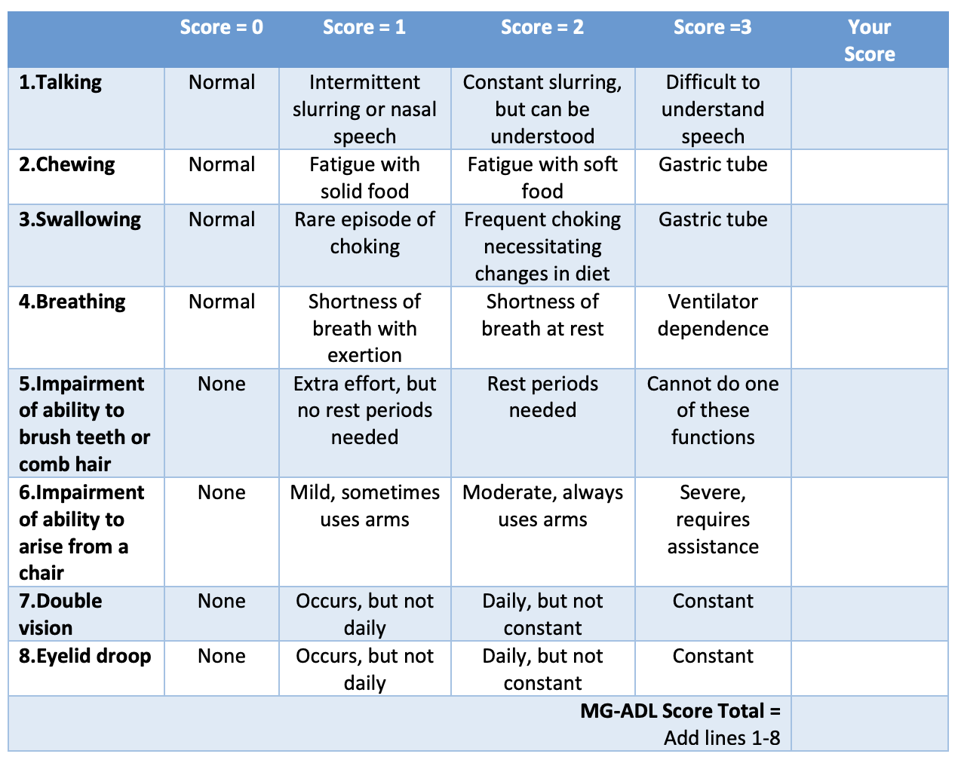
To accomplish this, 14 participants with generalized myasthenia gravis were recruited. All relied on immunosuppression drugs for their condition. Symptoms were graded from a scale of 0 to 36, and all participants had a score of six or higher (Table 1). They did not undergo a lymphodepletion regime, and most continued their previous medication course throughout the study (pyridostigmine for muscle weakness, and corticosteroids to suppress the immune system).
Table 1: Myasthenia Gravis Activities Daily Living (MG-ADL) scale. Symptom severity is assigned a score between 0 and 24 points. The participants in the study had a score of 6 or higher. CREDIT: Myasthenia Gravis of Foundation of Illinois
MYASTHENIA GRAVIS OF FOUNDATION OF ILLINOIS
The maximum tolerated dose was determined by giving three participants an escalating dose of mRNA-CAR T cells once a week, for three weeks. Once established, the other eleven participants were sorted into three groups with different treatment schedules:
- Two infusions weekly for three weeks,
- One infusion weekly for six weeks, or
- One infusion per month, for six months.
The intravenous infusion lasted 15 to 30 minutes each time, and the patients were monitored up to an hour afterwards.
Evaluations occurred on weeks 8, 12, 16 and 20, and then on months 6, 9 and 12. Changes in symptoms were measured using four different and validated scales for measuring myasthenia gravis severity.
Encouraging Results
Interestingly, the therapy did not produce any dose-limiting toxicities or treatment-related serious adverse events. As a result, the highest tested CAR T cell dose became the maximum tolerated dose by default.
One infusion per week, for six weeks, was established as the ideal dosing schedule. Participants in this group experienced the most significant and most prolonged decrease in disease severity; some experienced minimal symptom expression even months after the initial dosing regimen. It is encouraging to see that symptom reduction persisted up to 12 months for certain individuals.
The CAR T therapy also demonstrated a favorable safety profile. None of the patients experienced cytokine release syndrome or neurotoxicity, the two most common adverse effects associated with traditional CAR T therapy. The adverse events that did occur were resolved within 24 hours of the infusion. This included headache, nausea, vomiting and fever.
Future Implications
CAR T therapy is a needed alternative for many patients who do not respond to current cancer treatments. The platform, with certain adjustments, could also provide a safe and lasting reduction in symptoms for people who struggle with myasthenia gravis. The ability to target pathogenic plasma cells is especially novel in the realm of myasthenia gravis treatment. The authors posit that Descartes-08 may be most useful as an infrequent, as-needed treatment.
It is early days for mRNA based CAR T therapy to treat rare autoimmune diseases. This study demonstrates the promise for this prototypical therapy to accomplish what other treatments could not. It will be crucial to see how the therapy performs in the larger, randomized control trial currently in progress.