The most common form of ovarian cancer is also the most aggressive. These fast-growing cancer cells initially respond well to standard care—surgery, chemotherapy, monoclonal antibodies and drug inhibitors—but around 70% of patients experience relapse within three years of treatment. A new study turns to mRNA injections as a solution. Published in Cancer Communications, German researchers have discovered that an mRNA injection can rescue a gene that is often inactivated in patients with aggressive ovarian cancer activity in human tumor cells and in mice.
Aggressive Ovarian Cancer and Tumor Protein p53
Researchers at the Goethe-University Medical School in Germany sought to restore tumor-protective gene functions in people with high-grade serous ovarian cancer. They targeted tumor protein p53 (TP53), a gene which is mutated and nonfunctional in over 96% of aggressive ovarian cancer cases. This mutated gene is common in other human cancers and animal cancers, as well. Under normal conditions, this gene encodes a tumor-suppressing protein by the same name. The protein prevents cells with genome damage from replicating and encourages DNA repair; if the damage is irreparable, the protein induces the cell’s death to ensure that the faulty DNA is not propagated.
The team produced small pieces of genetic material called mRNA in the lab and delivered them to ovarian tumor cells using artificial vehicles called liposomes. Ideally, the tumor cells should read the synthetic mRNA and produce functional tumor protein p53 in cell culture and mice tests. If proven effective, this treatment could be adopted to treat other cancers that share the same mutated gene.
mRNA Injection Can Rescue Human Ovarian Cancer Cells in Culture
First, the study examines the effect of p53-mRNA on ovarian cancer cell cultures with mutated TP53 genes. Transfecting the cells with special mRNA resulted in cell cycle arrest and cell death, indicating normal p53 function and tumor suppression. Fascinatingly, the mRNA upregulates cell death genes that other developing therapeutics target (e.g. CDK1 and PLK1 checkpoint inhibitors). The team also discovered that the protein reprograms the cancer cells and increases the stability of mutant p53 through various mechanisms. The result suggests that p53-mRNA can reactivate some of the mutated gene’s functions and reduce the genetic instability of these cancer cells.
mRNA Injection Reduces Human Ovarian Tumor Cells in Mice
Next, the researchers explored the effects of treated ovarian cancer cells in mice. Cancer cells were transfected with either mock or p53-mRNA and then injected into the right ovary of each mouse. The team tracked the tumor growth in the mice for ten weeks and analyzed the organs afterward. The mRNA injection significantly slowed tumor growth in treated mice and prevented the tumor from spreading in the cavity surrounding the abdominal organs in a dose-dependent manner. Mock mice, in comparison, carried huge tumor masses on the ovaries and secondary tumors on nearby organs.
How does the treatment fare in vivo? To this end, mice received two injections of human ovarian cancer cells in their peritoneal cavity—the space around the organs in the abdomen. Hours later, p53 injections were administered through the same route. For the next three weeks, mice were either injected with two p53-mRNA injections per week or received a mock treatment.
The control mice formed weakly attached tumor masses on the surface of several organs. In contrast, the organs of the treated mice appeared normal and tumor-free.
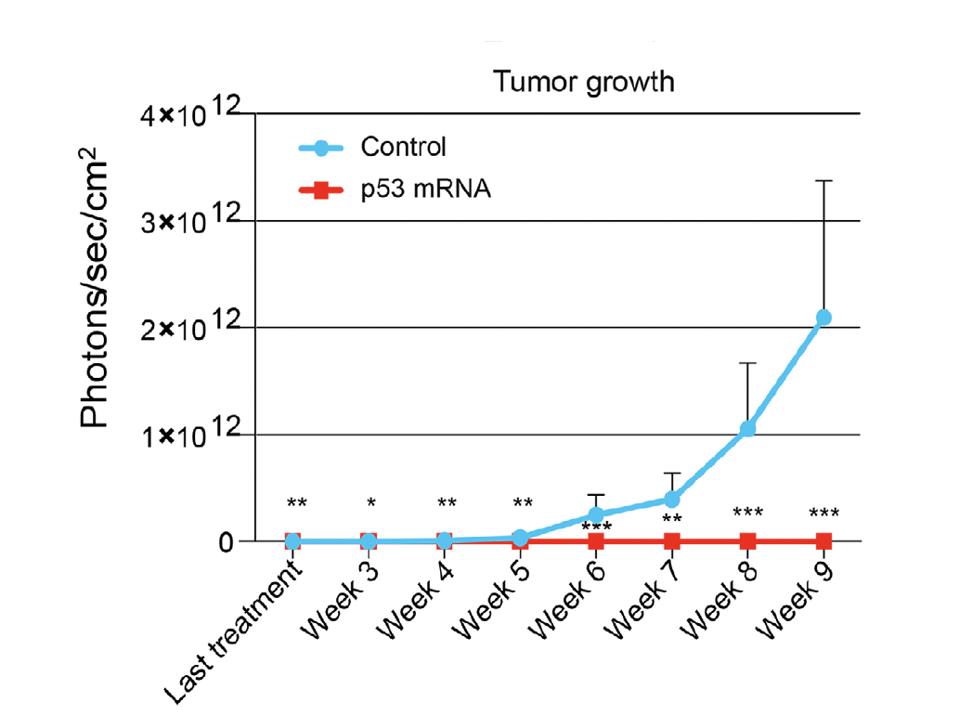
Repeated p53-mRNA injections significantly reduce tumor growth in treatment mice compared to controls.
FROM (2024) “RESCUE OF P53 FUNCTIONS BY IN VITRO-TRANSCRIBED MRNA IMPEDES THE GROWTH OF HIGH-GRADE SEROUS OVARIAN CANCER.” BY RAAB M, KOSTOVA ET AL.
Future Implications
It could be possible to treat aggressive ovarian cancer with an mRNA injection. As demonstrated in this study, mRNA injections can encourage healthy tumor protein p53 production in ovarian cancer cells and reactivate previously disabled tumor suppressive functions. The authors suggest that this effect could boost the effectiveness of carboplatin, a standard treatment for aggressive ovarian cancer. The study also underlines the crucial role tumor protein p53 plays in preventing DNA damage and, in turn, general cancer development.
This treatment, if proven effective in humans, promises to be accessible and relatively affordable to produce. mRNA manufacture relies on an enzymatic process called in vitro transcription, a process that does not require juggling live viruses or propagating living cells. The genetic material can therefore be produced quickly and at a large scale. Importantly, direct injection avoids lengthy and dangerous steps used in other cell therapies—mainly, extracting a patient’s cells and modifying them in the lab, and preparing the patient with immunosuppressing chemotherapy. This major advance could one day treat a wide variety of illnesses, including cardiac fibrosis and genetic blood disorders.


